Moderna, Inc.’s (MRNA) COVID-19 vaccine Spikevax has received full approval from the U.S. Food and Drug Administration (FDA) for treatment of the virus in individuals of ages 18 and above.
Following the news, shares jumped 6.2% to close at $169.33 on January 31. The biotechnology giant’s shares have lost almost 28% year-to-date vis-à-vis a 51.5% gain over the past year.
Full Approval for Spikevax
The FDA gave its full approval for Spikevax in adults based on the totality of scientific evidence submitted by the company. The evidence includes follow-up data from the Phase 3 COVE study, which demonstrated high efficiency and safety approximately six months after the second dose.
The FDA approved the Biologics License Application (BLA) for Spikevax, which earlier only had the Emergency Use Authorization (EUA) for prevention of the virus. The EUA meant that the company could not market the drug commercially as a product. Now, with full approval, the drug maker can commercialize the vaccine.
Moreover, the company also submitted plant and manufacturing data, which was required by the FDA for approval. The company’s COVID-19 vaccine has received approval from regulators in more than 70 countries including Canada, Japan, the European Union, the UK, and Israel. In some of these countries, usage of the vaccine in youth is approved.
Spikevax has been administered to millions worldwide and has been efficient in stopping hospitalizations and deaths, while protecting people from the deadly virus. In 2021, Moderna shipped about 807 million doses of the COVID-19 vaccine globally. Around 25% of these doses were shipped to low- and middle-income countries.
CEO Comments
CEO of Moderna, Stéphane Bancel, said, “The totality of real-world data and the full BLA for Spikevax in the United States rearms the importance of vaccination against this virus. This is a momentous milestone in Moderna’s history as it is our first product to achieve licensure in the U.S.”
“We are grateful to the U.S. FDA for their thorough review of our application. We are humbled by the role that Spikevax is playing to help end this pandemic,” Bancel concluded.
Analysts’ View
Responding to the news, Piper Sandler analyst Edward Tenthoff reiterated a Buy rating on the stock with a price target of $348, which implies a whopping 105.5% upside potential to current levels.
Tenthoff noted that the FDA’s full approval of Spikevax meant a broad de-risking of Moderna’s rich mRNA vaccine pipeline. Furthermore, the analyst stated that the drugmaker has advanced purchase agreements of $18.5 billion in 2022, which indicates promising future performance.
Additionally, the analyst is also encouraged by Moderna’s progress on the mRNA-1010 seasonal flu study.
Overall, the stock has a Moderate Buy consensus rating based on 6 Buys, 9 Holds, and 1 Sell. The average Moderna price target of $283.79 implies 67.6% upside potential to current levels.
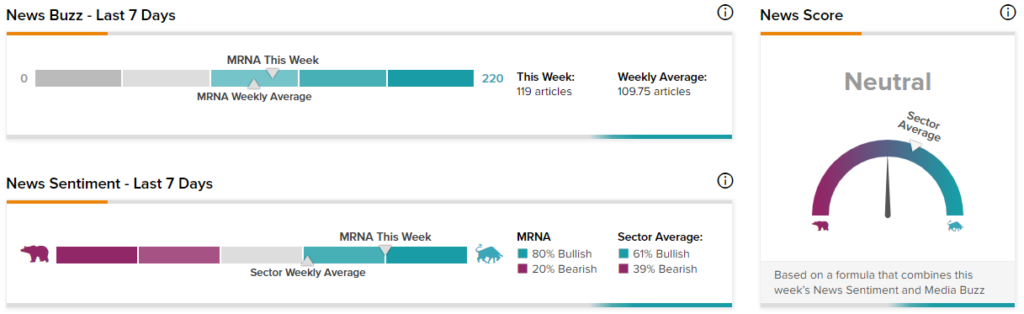
News Sentiment
TipRanks’ News Sentiment and Media Buzz Analysis shows the combined News Score based on MRNA’s News Sentiment and Media Buzz.
TipRanks data shows that the News Score for Moderna is currently Neutral based on 119 articles over the past seven days. 80% of the articles have a Bullish Sentiment compared to a sector average of 61%, while 20% of the articles have a Bearish Sentiment compared to a sector average of 39%.
Download the TipRanks mobile app now
To find good ideas for stocks trading at attractive valuations, visit TipRanks’ Best Stocks to Buy, a newly launched tool that unites all of TipRanks’ equity insights.
Read full Disclaimer & Disclosure
Related News:
Musk Criticizes Biden for Praising GM and Ford
Netflix CEO Hastings Buys $20M of Shares in the Dip
Caterpillar Drops 5% Despite Exceeding Q4 Expectations









