Akebia Therapeutics (AKBA) has announced the initiation of an investigator-sponsored study evaluating the use of vadadustat as a potential therapy to prevent and lessen the severity of acute respiratory distress syndrome (ARDS), a complication of COVID-19 infection.
The randomized, double-blind, placebo-controlled study is intended to evaluate the safety and efficacy of vadadustat in up to 300 adult patients who have been hospitalized at Memorial Hermann-Texas Medical Center for hypoxemia due to COVID-19.
Patients will be dosed with vadadustat or a placebo starting within 24 hours of hospital admission and continuing for up to 14 days. This study is being conducted by The University of Texas Health Science under a U.S. Food and Drug Administration Investigational New Drug (IND) application.
“Right now, we can support patients with COVID-related ARDS with supplemental oxygen and different forms of ventilation, but don’t yet have effective treatments to protect their lungs and help them get better,” said one of the principal investigators, Dr. Bentley J. Bobrow.
“Our goal is to prevent patients with the virus from progressing to requiring a ventilator and, if they do require a ventilator, to decrease the time they are on that ventilator.” ARDS can result in a rapid decline of respiratory function and death.
Akebia recently reported positive primary and key secondary efficacy and safety endpoint results from INNO2VATE, the first of its two global Phase 3 cardiovascular outcomes programs. Vadadustat is approved as a treatment for anemia due to chronic kidney disease in both dialysis-dependent and non-dialysis dependent adult patients in Japan, but not yet in the US.
An investigational oral HIF-PHI, vadadustat leverages the HIF mechanism and is designed to mimic the body’s physiologic response to low oxygen e.g. at high-altitudes. At higher altitudes, the body responds to lower oxygen availability with stabilization of HIF, which can lead to increased red blood cell production and improved oxygen delivery to tissues.
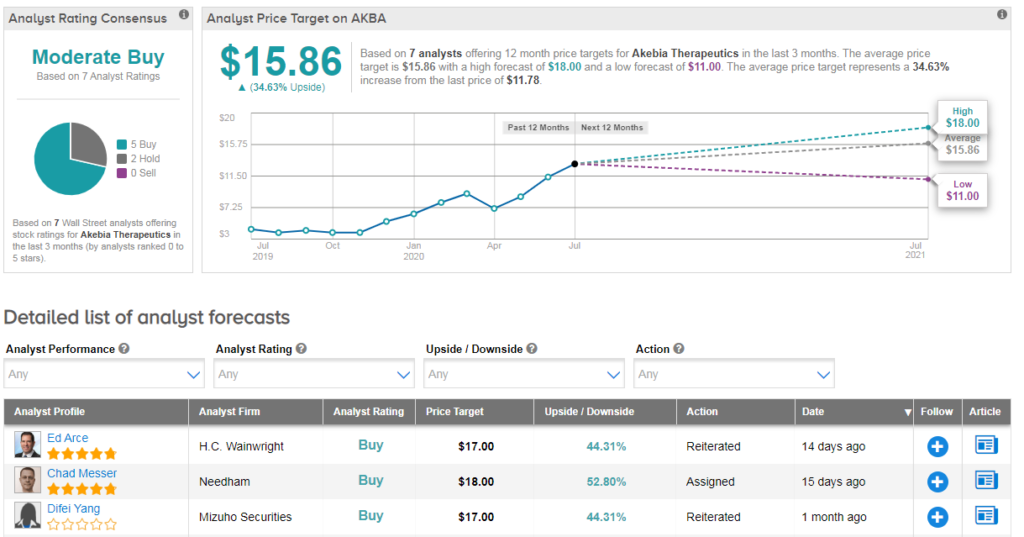
Shares in Akebia have surged 85% year-to-date, boosted by positive top-line results from the vadadustat trials. HC Wainwright analyst Ed Arce reiterated a buy rating on the stock and $17 price target (46% upside potential) following the recent Japan approval.
“With vadadustat’s first regulatory approval in hand, we continue to expect positive data from the Phase 3 PROTECT program to support vadadustat’s NDA [new drug application] filing around year end 2020, with its FDA approval and market launch around mid-2021 assuming the acquired Priority Review Voucher is utilized” commented Arce.
Overall analysts have a cautiously optimistic Moderate Buy consensus on Akebia with an average analyst price target of $16 (36% upside potential).(See AKBA stock analysis on TipRanks)
Related News:
Abbott Labs, Edwards Lifesciences Settle Heart Device Patent Disputes
Aldeyra Spikes 10% In Pre-Market On New Perceptive Stake
Equillium Explodes 260% On Positive Covid-19 Results; India Approval









