Verve Therapeutics (NASDAQ:VERV) shares plunged by nearly 33% in the early session today after the clinical-stage gene editing company halted patient enrollments in its Heart-1 Phase 1b trial of its lead candidate, VERVE-101.
Verve’s Setback
Verve focuses on developing single-course gene-editing medicines for cardiovascular diseases. Its lead candidate, VERVE-101, undergoes evaluation in patients with heterozygous familial hypercholesterolemia (HeFH), atherosclerotic cardiovascular disease, and uncontrolled hypercholesterolemia.
Thus far, Verve has administered doses to 13 subjects in the trial.
The decision to halt patient enrollments ensued due to a serious adverse event experienced by a subject within the first four days of dosing. The company is investigating the incident and intends to determine the trial’s next course of action following collaboration with regulatory authorities.
Verve’s Path Forward
The company now plans to prioritize the development of its second candidate, VERVE-102. It plans to begin the Heart-2 trial for the drug in HeFH in the second quarter of 2024. Additionally, the biotechnology company noted that it had sufficient cash balance to ensure a runway into late 2026.
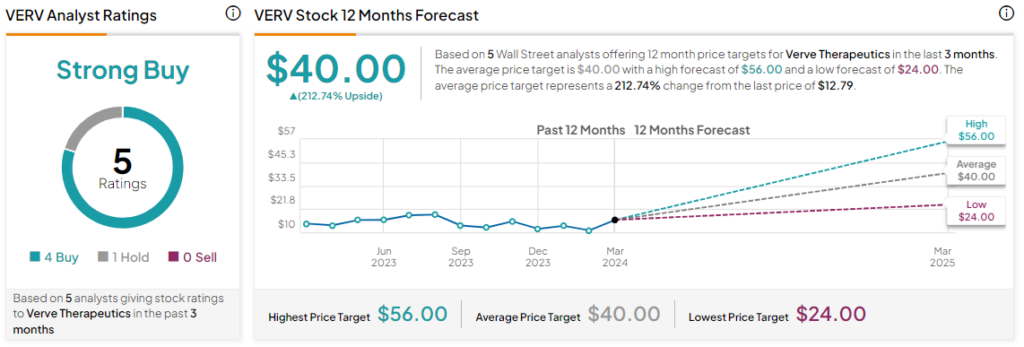
What Is the Price Prediction for Verve?
Today’s price decline further adds to the nearly 60% value erosion in Verve shares over the past three years. Overall, the Street has a Strong Buy consensus rating on the stock alongside an average VERV price target of $40.
Read full Disclosure









