Global pharmaceutical and biotechnology giant Pfizer (PFE) revealed that it has commenced patient dosing in its phase 3 clinical trial, TALAPRO-3, which will study talazoparib and enzalutamide in patients suffering from metastatic castration-sensitive prostate cancer (mCSPC).
The trial will evaluate the safety and efficacy of talazoparib, an oral poly (ADP-ribose) polymerase (PARP) inhibitor when dosed in combination with enzalutamide, an androgen receptor inhibitor. The trial will study 550 men with DNA damage response (DDR)-deficient mCSPC from 28 countries.
Chief Development Officer, Oncology, Pfizer Global Product Development, Chris Boshoff, said, “By combining enzalutamide, which has a proven clinical benefit in men with metastatic castration-sensitive prostate cancer, with talazoparib, our PARP inhibitor that is active in DDR-mutated cancer, we may be able to offer a new treatment option that targets the underlying genetic mechanisms associated with DDR-mutated mCSPC.”
Currently, Pfizer has received approval for talazoparib for treating adults suffering from deleterious or suspected deleterious germline BRCA-mutated (gBRCAm) HER2-negative locally advanced or metastatic breast cancer. (See Pfizer stock chart on TipRanks)
Argus Research analyst David Toung recently maintained a Hold rating on the stock with a price target of $55 (41% upside potential).
Toung said that while the market is underestimating the amount of revenue that the COVID-19 vaccine franchise will generate beyond 2021, Pfizer is boosting its production capacity to deliver billions of doses by the end of this year.
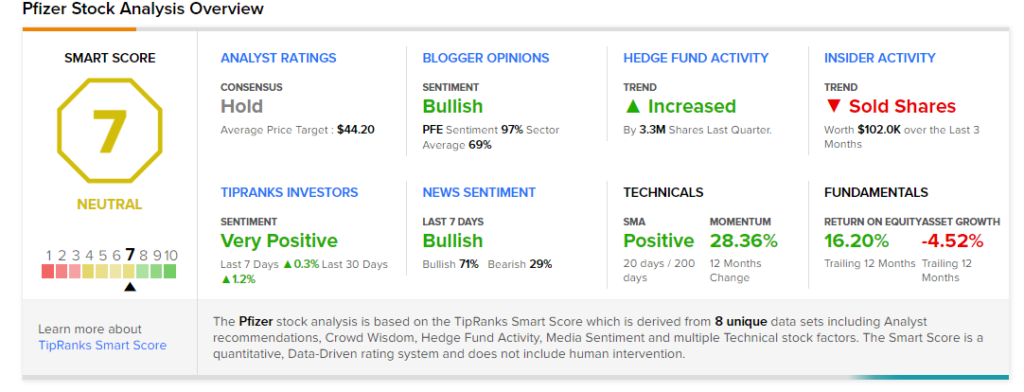
Overall, the stock has a Hold consensus rating based on 2 Buys, 9 Holds, and 1 Sell. The average Pfizer analyst price target of $44.20 implies 13.2% upside potential from current levels. The company’s shares have gained 28.1% over the past year.
According to TipRanks’ Smart Score rating system, Pfizer scores a 7 out of 10, suggesting that the stock is likely to perform in line with market averages.
Related News:
Winnebago Delivers Impressive Q3 Earnings, Revenues More Than Double
BeiGene’s Tislelizumab Gets Approval for First-Line Treatment of Advanced Non-Squamous NSCLC in China
Shake Shack to Deepen Presence in China; Shares Gain 1.5%









