Gilead Sciences Inc. (GILD) announced on Tuesday that it will buy a 49.9% equity stake in Pionyr Immunotherapeutics Inc., for $275 million.
Under the terms of the deal, Gilead has an exclusive option to purchase the remainder of Pionyr, a privately held company developing first-in-class cancer immunotherapies, for $315 million. In addition, Pionyr’s shareholders will be eligible to receive up to $1.47 billion in option exercise fees and future milestone payments.
“Pionyr is pursuing promising, novel biology in the field of immuno-oncology,” said Gilead’s Chairman and CEO Daniel O’Day. “The agreement represents important progress as we continue to build out Gilead’s presence in immuno-oncology with innovative and complementary approaches.”
Pionyr’s investigational cancer drugs, PY314 and PY159, have demonstrated preclinical efficacy, suggesting potential in solid tumors in combination with established anti-PD(L)-1 agents. Pionyr plans to file investigational new drug (IND) applications with the U.S. Food and Drug Administration (FDA) for both PY314 and PY159 in the third quarter of this year.
As part of the purchase agreement Gilead will provide Pionyr with additional funding for the PY314 and PY159 clinical programs, as well as ongoing research and development programs.
The announcement comes after Gilead said on Monday that it expects to produce more than 2 million treatment courses of its experimental coronavirus drug candidate remdesivir by the end of the year and many millions more in 2021.
Shares in Gilead declined less than 1% to $75.20 in afternoon U.S. trading trimming the year-to-date advance to about 16%.
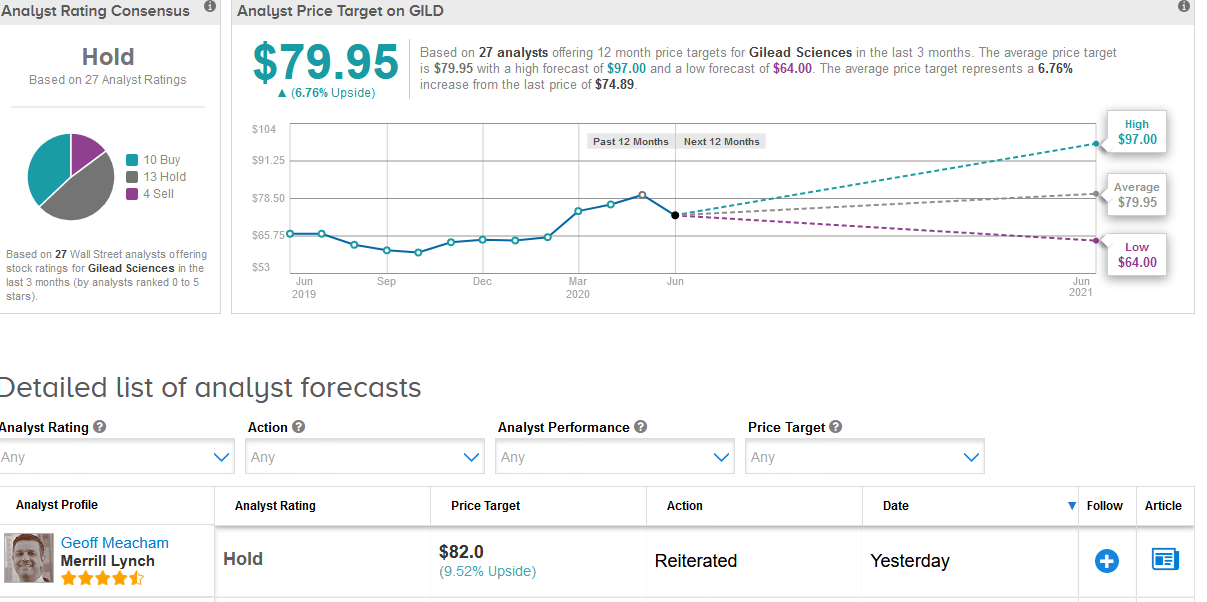
Wall Street analysts remain sidelined on the stock. The Hold consensus is based on 13 Hold ratings and 4 Sell ratings versus 10 Buy ratings. The $79.95 average price target implies 6.8% upside potential in the shares in the coming 12 months. (See Gilead stock analysis on TipRanks).
Related News:
Gilead Aims To Produce More Than 2 Million Remdesivir Courses By Year-End
Indian Drugmakers Get Regulatory Nod For Generic Version Of Gilead’s Remdesivir
5 Promising Covid-19 Vaccines Picked For Trump’s Operation Warp Speed