Drugmaker Eli Lilly & Co (LLY) said the U.S. Food and Drug Administration (FDA) approved its drug for lung and thyroid cancer treatment
Meet Your ETF AI Analyst
- Discover how TipRanks' ETF AI Analyst can help you make smarter investment decisions
- Explore ETFs TipRanks' users love and see what insights the ETF AI Analyst reveals about the ones you follow.
The drug selpercatinib, which will be sold under the name of Retevmo, was approved under the FDA’s Accelerated Approval regulations based on Phase 1/2 trial’s endpoints of objective response rate and duration of response.
Selpercatinib is used as an inhibitor for patients with advanced RET-driven lung and thyroid cancers. RET is a genetic mutation which leads to uncontrolled cell growth. The mutations have been found in about 2% of lung cancers and 10%-20% of papillary thyroid cancers.
“We are extremely proud of how quickly the combined Loxo Oncology and Lilly Oncology teams brought Retevmo to patients, further demonstrating our commitment to delivering life-changing medicines to people living with cancer,” said Anne White, president of Lilly Oncology. “Retevmo entered clinical trials in May of 2017 and is now approved less than three years later, representing the most rapid timeline in the development of an oncology medicine with multiple indications.”
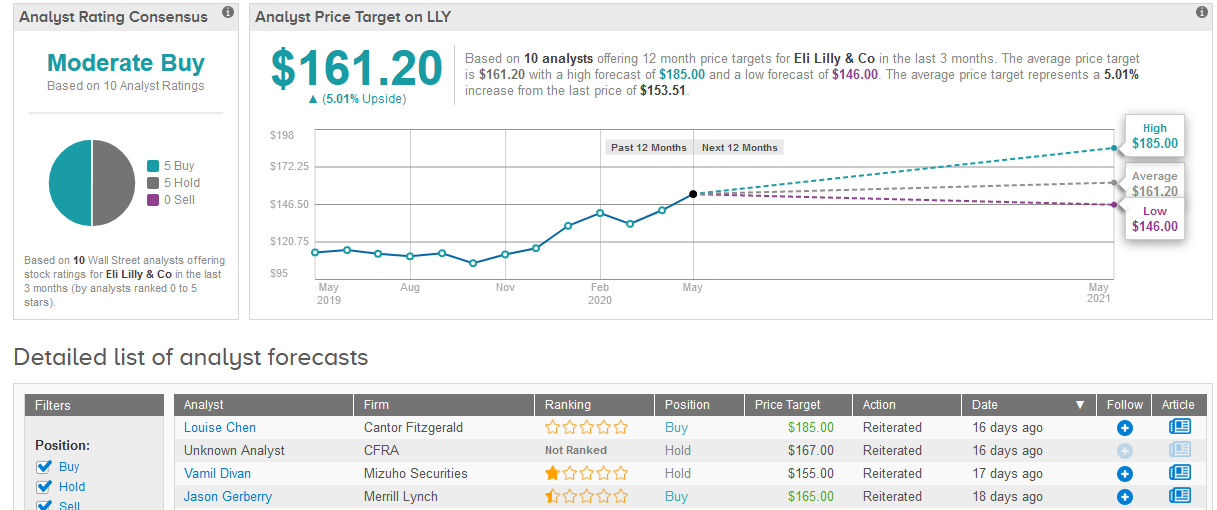
Shares in Eli Lilly have been on a winning streak since March 23, advancing 29% to $153.51 as of Friday.
Vamil Divan, analyst at Mizuho Securities at the end of last month maintained his Hold rating on the stock, while raising the price target to $155 from $148, saying that the investor bias towards safer, higher quality names will likely continue to support the shares.
“We believe the underlying fundamentals for the company remain strong,” Divan wrote in a note to investors. “Lilly’s current valuation appears stretched to us relative to its large cap biopharma peers so we maintain our Neutral rating.”
TipRanks data shows that Wall Street analysts are evenly divided on Eli Lilly’s stock between 5 Buys and 5 Holds adding up to a Moderate Buy consensus rating. The $161.20 average price target indicates upside potential of 5% in the coming 12 months. (See Eli Lilly’s stock analysis on TipRanks).
Related News:
AstraZeneca-Merck Ovarian Cancer Treatment Gets FDA Approval
Quidel’s Rapid Covid-19 Antigen Test Scores Emergency FDA Approval
Tesla’s Elon Musk Takes Legal Action to Fight Reopening of California Car Plant