Merck & Co. (MRK) on Tuesday became the latest drugmaker to join the race for the development of a COVID-19 treatment by announcing the purchase of Austrian vaccine maker Themis Bioscience and the collaboration with non-profit research group IAVI.
Shares in Merck rose 4% to $79.40 in U.S. pre-market trading. The drugmaker said that the Themis acquisition builds upon an ongoing collaboration between the two companies to develop vaccine candidates using the measles virus vector platform, and is expected to accelerate the development of Themis’ COVID-19 vaccine candidate. The vaccine candidate is in pre-clinical development, and clinical studies are planned to start later in 2020.
In addition, Merck announced a new collaboration with IAVI, a non-profit scientific research organization, to advance the development and global clinical evaluation of another vaccine candidate for the prevention of COVID-19.
This vaccine candidate will use the recombinant vesicular stomatitis virus (rVSV) technology that is the basis for Merck’s Ebola Zaire virus vaccine, ERVEBO (Ebola Zaire Vaccine, Live), which was the first rVSV vaccine approved for use in humans. The vaccine candidate is in preclinical development, and clinical studies are planned to start later this year. Merck will manage the regulatory filings globally.
Furthermore, Merck has also signed an agreement with the U.S. Biomedical Advanced Research and Development Authority (BARDA), to provide initial funding support for this effort.
“Merck is collaborating with organizations around the globe to develop anti-infectives and vaccines that aim to alleviate suffering caused by SARS-CoV-2 infection,” said Roger M. Perlmutter, President of Merck Research Laboratories. “Merck and IAVI are eager to combine our respective strengths to accelerate development of an rVSV vaccine candidate, with the goal of blunting the trajectory of the COVID-19 pandemic.”
Lastly, Merck and biotech company Ridgeback Bio announced a collaboration to advance the development of EIDD-2801, an oral antiviral candidate for COVID-19. Under terms of the agreement, Merck will get exclusive worldwide rights to develop and commercialize EIDD-2801. Ridgeback Bio will receive an undisclosed upfront payment, specified milestones and a share of EIDD-2801 net proceeds, if approved. Meanwhile, Merck will be in charge of clinical development, regulatory filings and manufacturing.
Shares in Merck appreciated some 15% in the past two months after losing about a third of their value this year.
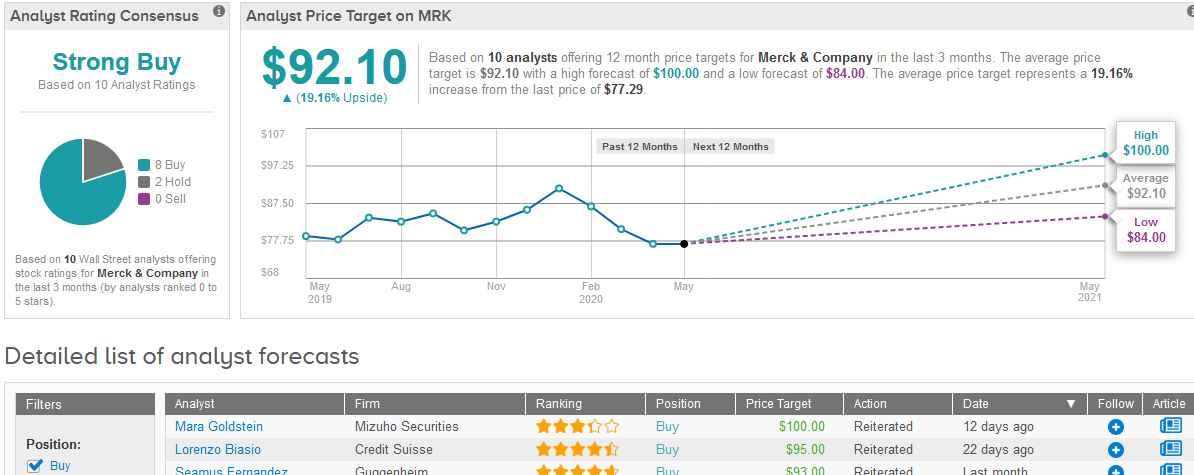
Following Merck’s announcements, Mizuho Securities analyst Mara Goldstein reiterated a Buy rating on the stock with a $100 price target.
“As a major player in vaccine development (also the first licensed Ebola vaccine), this move makes sense,” Goldstein wrote in a note to investors. “Given the scope of the global viral outbreak, we see room for multiple vaccine and therapeutic options.”
Overall, the stock has bullish support from the Street scoring 8 Buy ratings and 2 Hold ratings from analysts which makes the consensus a Strong Buy. The $92.10 average price target suggests 19% upside potential in the shares in the coming 12 months. (See Merck stock analysis on TipRanks).
Related News:
Novavax Begins Human Testing For Covid-19 Vaccine, Expects Results In July
Regeneron and Sanofi’s Dupixent Shows ‘Positive’ Trial Data, Meets Co-Primary Endpoints
Regeneron To Repurchase $5 Billion Stake From Sanofi