Corvus Pharmaceuticals Inc. (CRVS) shares shot up 115% on Tuesday after the clinical-stage biopharma company said that it has initiated a Phase 1 study to investigate a novel immunotherapy approach for patients with COVID-19.
The stock surged to $5.85 in early morning trading. Novavax reported that the first cohort of 5 patients enrolled in the study was treated at Temple University Hospital in Philadelphia. The initiation follows the U.S. Food and Drug Administration’s (FDA) review and approval of the company’s investigational new drug (IND) application for the COVID-19 study. The trial will evaluate anti-viral antibody response in up to 30 COVID-19 patients with mild to moderate symptoms.
Corvus is studying an agonistic (immunostimulatory) humanized monoclonal antibody, CPI-006, which has demonstrated a potential new approach to immunotherapy of infectious diseases and cancer. In both in vitro and in vivo studies in cancer patients, CPI-006 has demonstrated binding to various immune cells and the inducement of a humoral adaptive immune response leading to the production of antigen-specific immunoglobulin antibodies. The use of CPI-006 has also led to increased levels of memory B cells, which are the cells responsible for long-term immunity.
Corvus reported that a similar production of antibodies and memory cells to pathogens such as severe acute respiratory syndrome coronavirus 2, the virus that causes COVID-19, may provide immediate and long-term clinical benefits for patients including shortened recovery time and improved long-term protective immunity.
“Our monoclonal antibody may be a potential immunotherapy for COVID-19 based on its ability to stimulate the production of anti-SARS-CoV-2 antibodies,” said Corvus CEO Richard A. Miller. “We believe that COVID-19 patients treated with CPI-006 may benefit from an improved time to recovery and building longer term immunity. We believe this opens an entirely new area of investigation and opportunities to both treat and prevent serious infectious diseases.”
Miller added that the COVID-19 study broadens the company’s pipeline as it continues to advance its core cancer programs with ciforadenant and CPI-006, while remaining on track to provide data updates at medical meetings later this year.
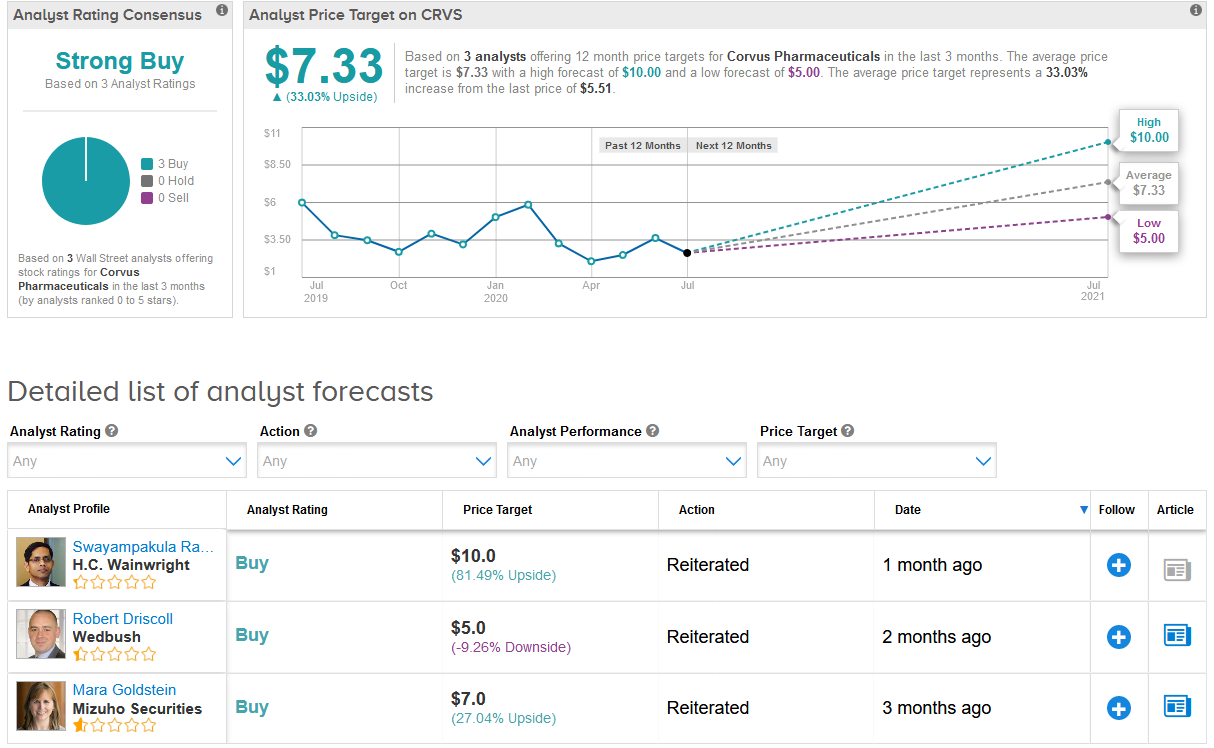
Shares in Corvus have slumped 50% year-to-date as of yesterday with the $7.33 average price target set by analysts implying shares are poised to gain 33% over the coming year. (See Corvus stock analysis on TipRanks)
Mizuho Securities analyst Mara Goldstein, who has a Buy rating on the stock with a $7 price target, says she likes the shares “based on the view that data can reset the valuation, and the 1Q20 update call identified progress that could lead to this in 2020.”
The rest of the Street is also bullish on the stock assigning only Buy ratings adding up to a Strong Buy consensus.
Related News:
Novavax Spikes 42% Pre-Market On $1.6B U.S. Funding For Covid-19 Candidate
Cellectis Sinks 13% In Extended Trading After FDA Halts Cancer Clinical Trial
Chembio Gains 12% After-Hours On New Covid-19 BARDA Contract