Robert Califf, Commissioner at the U.S. Food and Drug Administration (FDA), has indicated that the agency’s food safety division needs to undergo reforms following its delay in responding to a whistleblower report about safety lapses at Abbott Laboratories’ (NYSE: ABT) baby formula plant in Michigan.
Meet Your ETF AI Analyst
- Discover how TipRanks' ETF AI Analyst can help you make smarter investment decisions
- Explore ETFs TipRanks' users love and see what insights the ETF AI Analyst reveals about the ones you follow.
He blamed the division’s slow coordination and use of outdated technology as reasons behind the delay.
“These problems that we’re describing are systemic and they weren’t fixed. We definitely need a stronger team at the high levels on the food side as part of a revamping of the entire food program,” Califf said.
Meanwhile, the FDA has added the launch of untested baby formula to Abbott’s existing list of lapses. The issue was mentioned in a report released last month.
Earlier this year, the regulatory body had identified safety lapses, such as standing water on the floor and the presence of dangerous bacteria, during its inspection of the facility. Following the FDA’s findings, the company recalled a few products and shuttered the plant in February.
Califf said, “Frankly the inspection results were shocking. We had no confidence in the integrity of the Abbott quality program at this facility.”
However, he added that the healthcare giant was taking appropriate steps to reopen the plant by next month.
Last month, Abbott reported higher-than-expected earnings for the first quarter of 2022. EPS of $1.73 surpassed the Street’s estimate of $1.47 and the year-ago figure of $1.32.
Stock Rating
Based on seven Buys and one Hold, the stock has a Strong Buy consensus rating. ABT’s average price target of $139.38 implies 23.1% upside potential from current levels. Shares have lost 9.4% over the past six months.
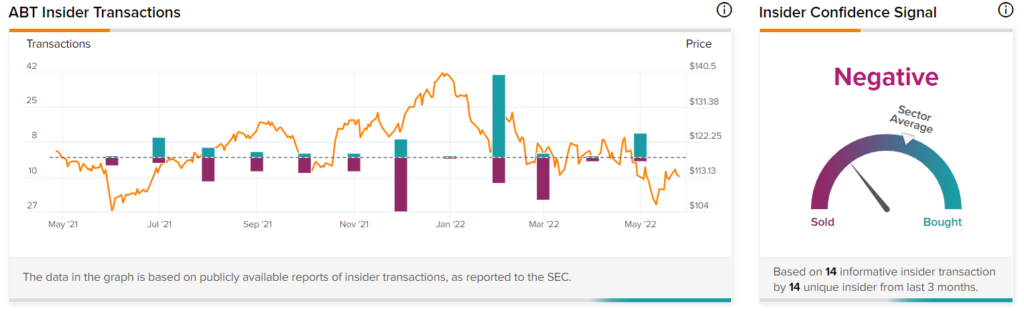
Insider Trading Activity
TipRanks’ Insider Trading Activity tool shows that Insider Confidence is currently Negative on Abbott. In the last three months, corporate insiders have sold $8.3 million worth of the company’s shares.
Conclusion
The reopening of Abbott’s Sturgis plant next month could bring some relief to the issue of baby formula shortage, which was exacerbated by the shutting down of the facility. It will also revive the company’s image, which has been tarnished following the FDA’s revelation of safety lapses at the facility.
Read full Disclosure
















