Shares in Pfizer Inc. (PFE) dropped 6% in after-market trading on Friday after the company reported a disappointing outcome for its Phase 3 PALLAS early breast cancer study.
Elevate Your Investing Strategy:
- Take advantage of TipRanks Premium at 55% off! Unlock powerful investing tools, advanced data, and expert analyst insights to help you invest with confidence.
Following a preplanned efficacy and futility analysis, the independent Data Monitoring Committee determined that the trial of Ibrance (palbociclib) plus standard adjuvant endocrine therapy is unlikely to show a statistically significant improvement in the primary endpoint of invasive disease-free survival (iDFS).
Patients receiving palbociclib in the study will be advised about next steps by their physicians and long-term follow up will proceed as planned, the company said, adding that no unexpected new safety signals were observed in patients receiving palbociclib.
The PALLAS trial compared palbociclib plus standard adjuvant endocrine therapy to standard adjuvant endocrine therapy alone in women and men with hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-) early (stage 2 and 3) breast cancer.
“We are disappointed in this outcome. Breast cancer is a leading cause of death around the world and delaying or preventing the development of metastatic disease is a significant unmet need. PALLAS is a large study with many subgroups and we are actively collaborating to determine if there are patients who may benefit from adjuvant treatment with the palbociclib combination,” said Chris Boshoff of Pfizer Global Product Development.
Meanwhile Pfizer CEO Albert Bourla moved to reassure investors, writing that the company’s growth projections are not reliant upon any individual pipeline opportunity. “We remain highly confident in our ability to deliver… a compound annual growth rate for revenues of at least 6% through 2025” he stated.
The full results from the PALLAS study will be shared at a later date. Palbociclib is also being studied in patients with high-risk early breast cancer and results from the collaborative PENELOPE-B trial are expected later this year. In the US, Ibrance is already approved for adult patients with HR+, HER2- advanced or metastatic breast cancer.
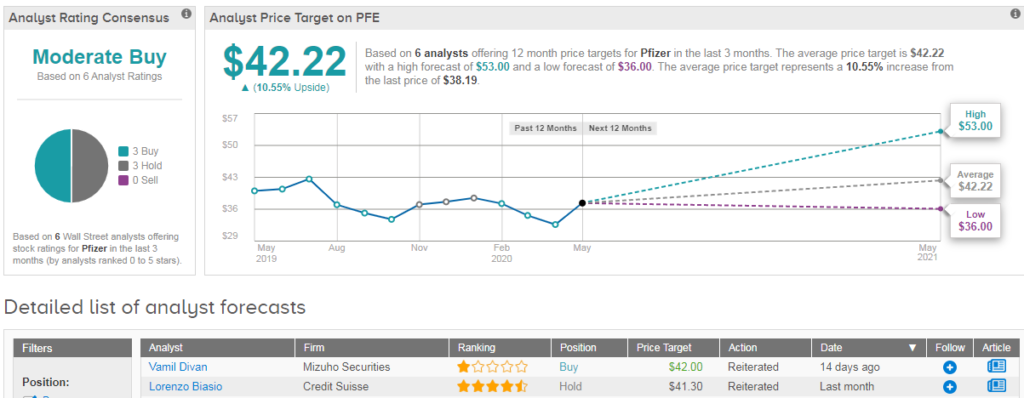
Shares in Pfizer are trading down 3% on a year-to-date basis, and analysts are evenly divided between hold and buy. This gives the stock a Moderate Buy Street consensus with an average price target of $42 (11% upside potential). (See Pfizer stock analysis on TipRanks).
Related News:
BioMarin Provides Positive Gene Therapy Update For Severe Hemophilia A
Efgartigimod’s Positive Data Is Good News for Momenta’s Nipocalimab
Novavax Seeks To Make 1 Billion Covid-19 Vaccine Doses
















