Pfizer and German partner BioNTech (BNTX) have won another order from the European Union to supply an additional 100 million doses of its Comirnaty COVID-19 vaccine in 2021.
The announcement came as the European Commission decided to exercise its option to purchase an additional 100 million doses under its Advanced Purchase Agreement signed with Pfizer (PFE) and BioNTech on Nov. 11. With the latest order, the total number of doses to be delivered to the 27 European Union member states stands at 300 million.
Pfizer and BioNTech’s COVID-19 vaccine will be produced in these two companies’ manufacturing sites in Europe. On Dec. 21, Pfizer announced that the European Commission granted a conditional marketing authorization (CMA) for Comirnaty (also known as BNT162b2), for active immunization to prevent COVID-19 in individuals 16 years of age and older.
The vaccine has now been granted conditional marketing authorization, emergency use authorization, or temporary authorization in over 40 countries globally, including all 27 EU member states. (See PFE stock analysis on TipRanks)
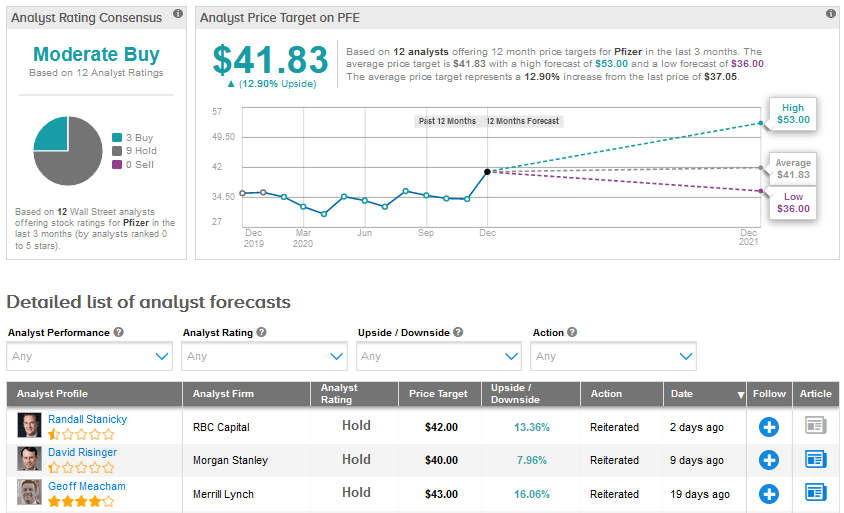
On Dec. 16, RBC Capital analyst Randall Stanicky downgraded Pfizer to Hold from Buy and lowered the price target to $42 from $43. Stanicky stated in a research note to investors, “What PFE has accomplished in getting a vaccine to market in record time, without taking government money, is nothing short of remarkable. But we believe the ‘stock’ reflects the vaccine DCF opportunity, making it harder for us to argue for more meaningful upside to our price target.”
“There is still much we don’t know, but that also includes the level of vaccine competition that could catch up in 2021,” added Stanicky.
Meanwhile, the Street has a cautiously optimistic outlook on Pfizer, with a Moderate Buy analyst consensus based on 3 Buys and 9 Holds. Shares appear fully priced currently and the average price target of $41.83 implies an upside potential of 12.9% over the coming year.
Related News:
Immutep Secures US Patent For Cancer Drug; Shares Jump 9.5%
Quanterix Wins FDA Approval For Covid-19 Antibody Test; Shares Rise 6%
Arcturus Gets Approval For Covid-19 Vaccine Phase 2 Study; Shares Plunge 38%