Scientists and government sources claimed today that Johnson & Johnson’s single-jab COVID-19 vaccine could be given emergency use authorization as early as next month, according to a report in the Daily Mail.
The Johnson & Johnson (JNJ) vaccine tricks the human body into producing antigens that are normally found on the coronavirus’ surface by using a harmless adenovirus to deliver genetic material, the Daily Mail reports, adding that the JNJ vaccine can be stored and transported in normal fridges, similar to the Oxford vaccine.
The Daily Mail has confirmed that Britain already ordered 30 million doses of the vaccine and has the option of ordering 22 million more, which would boost the mass immunization program and help the government fulfill its ambitions of vaccinating 13 million Britons by the middle of February.
Commenting on the efficacy of the vaccine, Sir John Bell, professor of medicine at the University of Oxford and an adviser to the U.K.’s Vaccine Taskforce, told the Daily Telegraph that he believes, “it’ll work well,” adding that he thinks “it may well have a material impact on what we can do in the UK to get more people vaccinated.”
Johnson and Johnson said that as soon as the vaccine gets the required approvals, it is ready to ship doses from its factories in Europe. (See JNJ stock analysis on TipRanks)
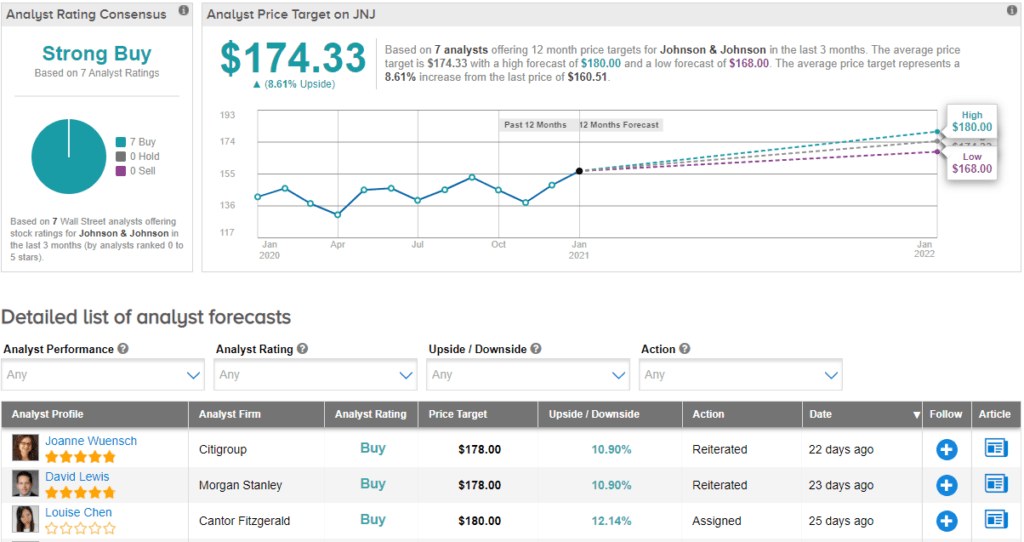
Morgan Stanley analyst David Lewis reiterated his Buy rating on JNJ three weeks ago and maintained his price target of $170. This implies upside potential of around 11% from current levels.
Commenting after JNJ’s initial announcement in September that it had begun its large-scale Phase 3 trial of its COVID-19 vaccine, Lewis explained that his price target was based on a 19x multiple off his base case 2021 expected EPS, and in his view, “litigation liability has been more than reflected in J&J shares, creating a meaningful valuation disconnect vs. the S&P.”
Consensus among analysts is a Strong Buy based on 7 Buy ratings. The average price target of $174.33 suggests around 9% upside potential over the next 12 months.
Related News:
Nvidia Being Investigated By Regulators for $40B Arm Deal
Greenbrier Posts Wider-Than-Expected 1Q Loss; Top Analyst Says Buy
T-Mobile Expands 5G Coverage; Street is Bullish




















