NuVasive announced that the US Food and Drug Administration (FDA) has approved the NuVasive Simplify Cervical Artificial Disc (Simplify Disc) for two-level cervical total disc replacement (cTDR). Shares of the spine technology innovator rose 2.1% to close at $66.65 on April 6.
The US regulator’s decision was based on a two-level FDA Investigational Device Exemption (IDE) study which demonstrated that the Simplify Disc has an overall success rate of 10% higher than anterior cervical discectomy and fusion (ACDF). Furthermore, the device has the highest overall clinical success rate compared to any other approved cervical disc at both one- and two-levels, the company said.
The Simplify Disc, part of the NuVasive (NUVA) C360 portfolio, offers broad and systematic integrated solutions for the cervical spine across ACDF, posterior cervical fusion, and cTDR procedures.
NuVasive’s Global Business Units EVP Massimo Calafiore said, “This approval is an incredible achievement for the Simplify Disc and further broadens the growth opportunities for the NuVasive C360 portfolio. There is immense surgeon excitement for this latest edition to our cervical offering, and the expanded two-level indication provides greater opportunities to bring the most clinically effective technology in the cTDR market to more patients.” (See NuVasive stock analysis on TipRanks)
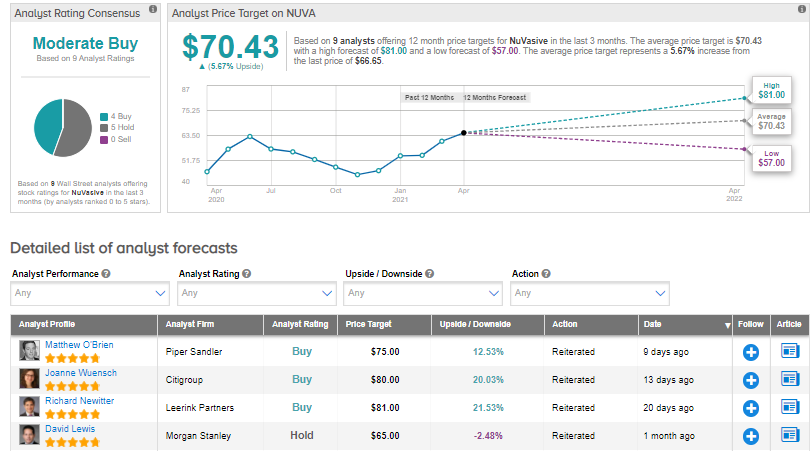
On March 25, Citigroup analyst Joanne Wuensch lifted the stock’s price target to $80 (20% upside potential) from $68 and reiterated a Buy rating.
In a note to investors, Wuensch said that NuVasive’s acquisition of Simplify Medical will expand its reach in the artificial cervical disc replacement market.
The rest of the Street is cautiously optimistic about the stock with a Moderate Buy consensus rating. That’s based on 4 analysts suggesting a Buy and 5 analysts recommending a Hold. The average analyst price target of $70.43 implies 5.7% upside potential to current levels. Shares have gained 32% over the past year.
Related News:
ViacomCBS’ VCNI To Buy Chilevision; Street Sees 40% Upside
FDA Rejects Acadia’s Supplemental New Drug Application For NUPLAZID; Shares Plunge
United Therapeutics Receives FDA Approval For Tyvaso Therapy; Shares Pop 15%




















