Shares of interventional oncology company Delcath Systems (NASDAQ:DCTH) are up a massive 95% at the time of writing after the U.S. Food and Drug Administration (FDA) gave approval to its Hepzato kit for the treatment of unresectable hepatic-dominant metastatic uveal melanoma (mUM).
Elevate Your Investing Strategy:
- Take advantage of TipRanks Premium at 50% off! Unlock powerful investing tools, advanced data, and expert analyst insights to help you invest with confidence.
The approval, as a liver-directed treatment, makes Hepzato the only approved liver-directed therapy on the market for this indication. mUM is a rare and aggressive metastatic cancer, with 90% of the cases involving the liver. The disease has an incidence rate of ~1,000 cases every year in the U.S.
The company now plans to partner with cancer centers across the U.S. to set up a network of treatment sites, with an anticipated commercial product launch in the fourth quarter. Additionally, Delcath plans to expand the development of the treatment to other tumor types.
Importantly, this FDA approval also triggers the second financing tranche under a PIPE (private investment in public equity) mechanism from March this year. The company estimates that it will realize $34.9 million in additional funding as a result.
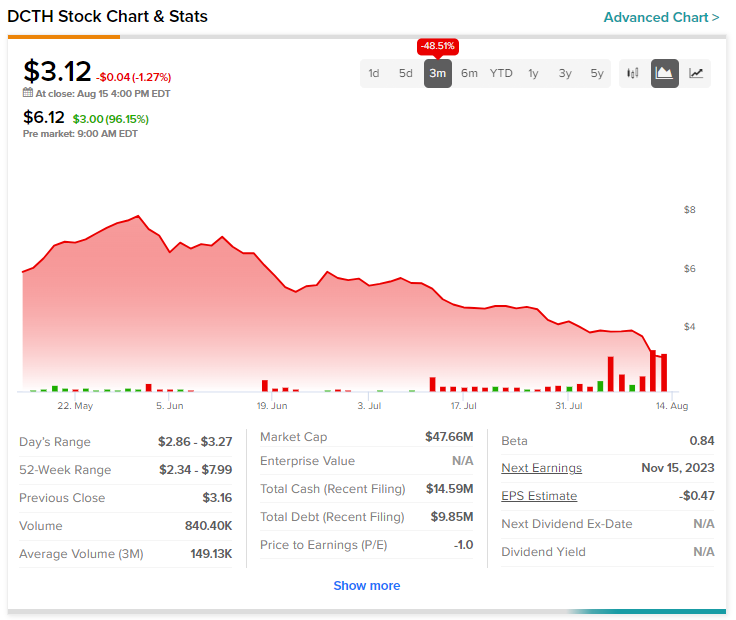
Today’s price gains come after a nearly 48.5% slide in Delcath shares over the past three months. In the meantime, the Street has a $17 consensus price target on the stock, pointing to a more than 400% potential upside.
Read full Disclosure
















