Shares of Acadia Pharmaceuticals (ACAD) gained over 15% on June 15 to close at $18.77 after the Food and Drug Administration (FDA) posted briefing documents for pimavanserin for treatment of hallucinations and delusions associated with Alzheimer’s disease psychosis (ADP).
Headquartered in California, Acadia Pharmaceuticals is a biopharmaceutical company that focuses on the development and commercialization of small-molecule drugs that address unmet medical needs in central nervous system disorders.
Details
The company’s lead candidate, NUPLAZID (pimavanserin), is aimed at the treatment of hallucinations and delusions associated with Parkinson’s disease psychosis. It is under phase 3 development for the treatment of Alzheimer’s disease psychosis and negative symptoms of schizophrenia.
A group of independent experts will meet with the FDA to discuss and advise on the approvability of pimavanserin for the treatment of hallucinations and delusions targeted at patients with Alzheimer’s disease.
Though the FDA is not under any obligation to follow the recommendations of its advisory committees, it usually does so.
Earlier in 2021, the FDA rejected pimavanserin in 2021 for Dementia-related psychosis (DRP). Following the rejection, Acadia resubmitted a supplemental New Drug Application in February targeting ADP.
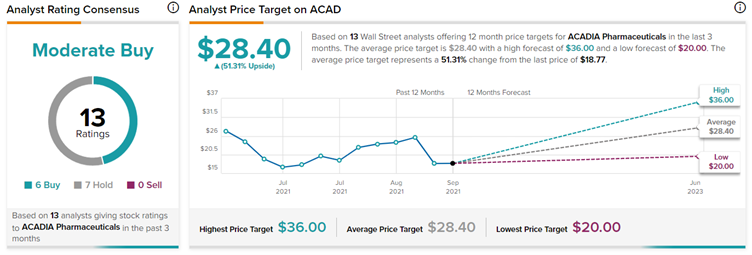
Wall Street’s Take
Following the FDA’s documents release, H.C. Wainwright analyst Andrew Fein reiterated a Buy rating on Acadia Pharmaceuticals with a price target of $36 (91.8% upside potential).
According to Fein, the recent development opens a “window of opportunity” for Acadia to argue for approval of pimavanserin in Alzheimer’s disease psychosis.
The Wall Street community is cautiously optimistic about the stock, with a Moderate Buy consensus rating based on six Buys and seven Holds. The average ACADIA Pharmaceuticals price target of $28.40 implies a 51.31% upside potential to current levels.
Conclusion
For a long time, FDA’s stance was skeptical with regards to the efficacy data supporting approval in ADP. However, this time, there was no major negative surprise coming out of the briefing document.
If ACAD is successful in convincing the panel tomorrow regarding the efficacy of the drug and wins a subsequent approval, it may unlock significant value for the stock.









