The U.S. Centers for Disease Control and Prevention (CDC) is backing the U.S. Food and Drug Administration’s (FDA) full approval of Moderna Inc.’s (NASDAQ: MRNA) COVID-19 vaccine Spikevax, according to Reuters.
Following the news, shares of the global pharmaceutical giant rose as much as 2.2% during intraday trading before ending the day almost flat at $163.01 on February 4.
Full Approval from U.S. CDC
Rochelle Walensky, Director of the U.S. CDC, has signed off in support of the FDA’s full approval for Moderna’s Spikevax for adults aged 18 years and above. The vaccine was earlier approved only under Emergency Use Authorization since December 2020. The CDC’s approval was the last formal consent required to make Spikevax a commercial vaccine.
The FDA approves the vaccines and the CDC signs off on the application and usage of the drug in the U.S. Around 75 million individuals in the U.S. have been fully vaccinated with Moderna’s vaccine.
Spikevax becomes the second drug after Pfizer Inc. (PFE) and BioNTech SE (BNTX) the COVID-19 vaccine received full approval for individuals aged 16 and above in August 2021.
Consensus View
The MRNA stock has a Moderate Buy consensus rating based on 5 Buys, 9 Holds, and 1 Sell. The average Moderna price target of $275.17 implies 68.8% upside potential to current levels. MRNA shares have lost 30.6% year-to-date, vis-s-vis gaining 44.6% over the past year.
News Sentiment
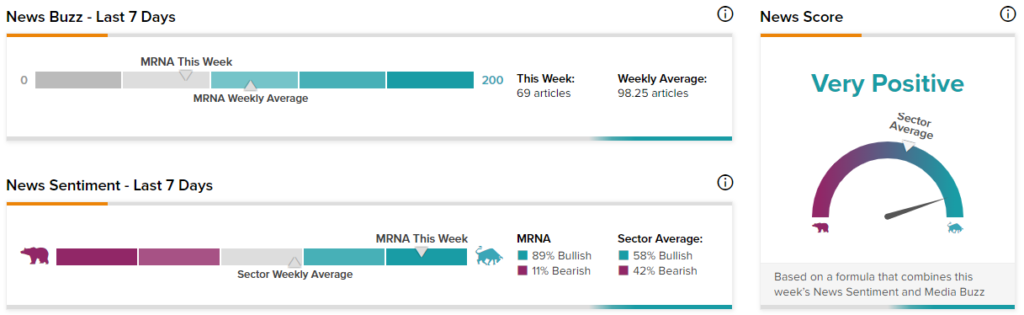
TipRanks data shows that the News Score for Moderna is currently Very Positive based on 69 articles over the past seven days. 89% of the articles have a Bullish Sentiment compared to a sector average of 58%, while 11% of the articles have a Bearish Sentiment compared to a sector average of 42%.
Download the TipRanks mobile app now
To find good ideas for stocks trading at attractive valuations, visit TipRanks’ Best Stocks to Buy, a newly launched tool that unites all of TipRanks’ equity insights.
Read full Disclaimer & Disclosure
Related News:
Snap Delivers Stellar Q4 Results; Shares Skyrocket 59% After Hours
Biogen Delivers Mixed Q4 Results; Shares Hit All-Time Low
Eli Lilly Drops 2.4% Despite Beating Q4 Expectations









