Square has expanded to Ireland. The payments solutions provider will offer an Early Access Programme on a first-come first-serve basis that will offer exclusive access to “an integrated suite of tools to seamlessly sell in-person and online” to small and medium enterprises (SMEs) in Ireland
Square (SQ) Europe’s Executive Director, Jason Lalor said, “We’re excited to bring Square’s remarkable solutions to Irish businesses. We look forward to partnering with Ireland’s rich community of independent business owners to refine and perfect our products in advance of launch, ensuring that they offer the time and cost savings that will enable Irish businesses to grow and prosper both in-person and online.”
There will be no long-term contracts or start-up fees for the Early Access Programme and the company expects its products to be generally available across Ireland later this year. The company’s integrated suite of products enables merchants to set up shops online, manage inventories and accept card payments in person or using smartphone and tablets. (See Square stock analysis on TipRanks)
On April 7, Barclays analyst Ramsey El-Assal raised the price target from $320 to $330 and reiterated a Buy rating on the shares. El-Assal said in a research note that the company is “finally positioned correctly” to realize its full potential as it has increased its business investment significantly outside the United States.
Shares of SQ have rallied 12.6% in the past five days.
Overall, the rest of the Street is cautiously optimistic on the stock with a Moderate Buy consensus rating based on 20 Buys, 10 Holds and 3 Sells. The average analyst price target of $276.67 implies that SQ shares have 7.1% upside potential to current levels.
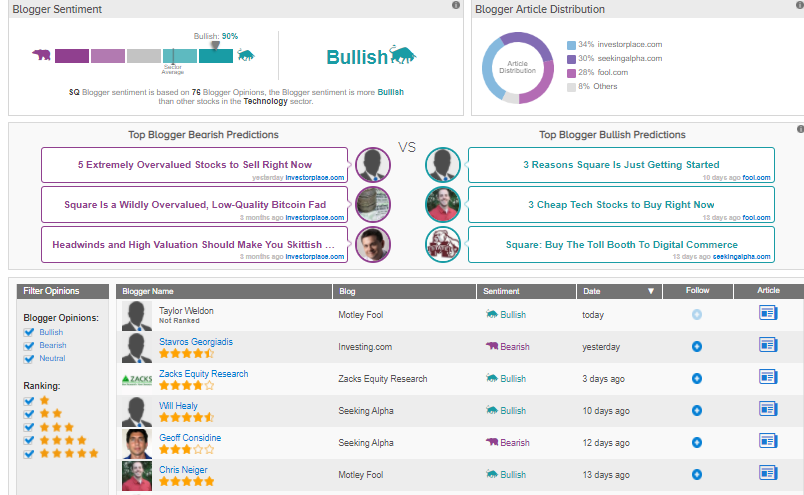
According to TipRanks data, financial bloggers‘ opinions are 90% bullish on SQ.
Related News:
Tyson Foods Officially Opens New $425M Tennessee Poultry Complex
Levi’s 1Q Sales And EPS Beat Estimates; Street Says Buy
Provention Bio Shares Crash 40% Due To Regulatory Setback For Diabetes Drug









