The U.S. Department of Health and Human Services (HHS) and the Department of Defense (DOD) have signed an agreement with New York-based biotechnology company Regeneron (REGN) to buy 1.4 million additional doses of REGEN-COV (casirivimab and imdevimab).
The U.S. Food and Drug Administration (FDA) has given an emergency use authorization to REGEN-COV to treat people, who have been infected by COVID-19 or are in certain post-exposure prophylaxis settings. (See Regeneron stock chart on TipRanks)
Following the announcement of the news, Regeneron’s shares gained 1.7% in extended trade on Tuesday and closed at $656. The company is engaged in the discovery, development, production and sale of medicines for the treatment of various medical conditions. Its offerings include ZALTRAP, ARCALYST, Libtayo, Kevzara, Praluent, Dupixent and EYLEA.
The President and CEO of Regeneron, Leonard S. Schleifer, said, “While vaccination remains the first line of defense to decrease the burden of COVID-19, REGEN-COV is a key tool that reduces the risk of hospitalization or death by 70% in high-risk individuals when given early in the course of the infection.”
As per the agreement, the company will provide 1,200 mg doses of REGEN-COV to the U.S. government by January 31, 2022, and charge $2,100 for each dose. This is Regeneron’s third agreement with the U.S. government. The first two were announced in July 2020 and January 2021.
The company has partnered with Roche Holding (RHHBY) for the development and distribution of REGEN-COV outside the U.S.
On September 9, UBS analyst Esther Rajavelu initiated coverage on the stock with a price target of $763 (18.3% upside potential). The analyst said, “The company should be able to generate strong topline growth with diversification but its oncology pipeline might take longer to create more value.”
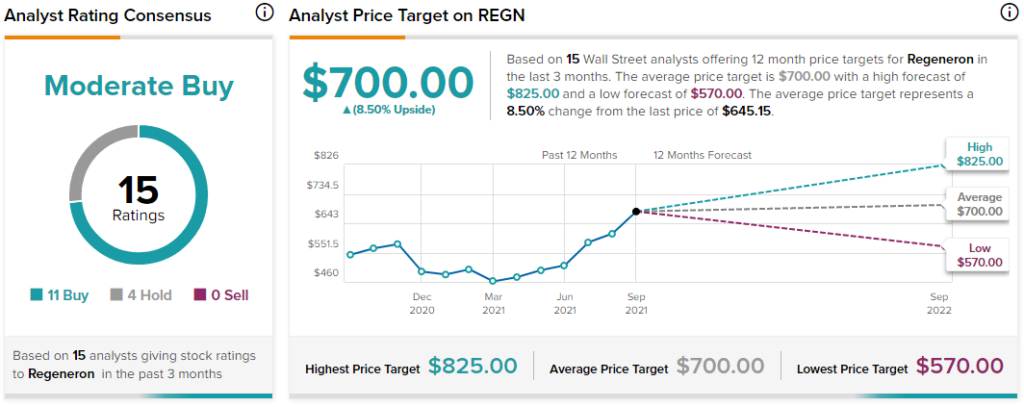
Overall, the stock has a Moderate Buy consensus rating based on 11 Buys and 4 Holds. The average Regeneron price target of $700 implies 8.5% upside potential. The company’s shares have gained 33.6% year-to-date.
Related News:
Arthur J. Gallagher Snaps Up Lloyd Bedford Cox, Impresses at Investor Day
A Look at FS KKR Capital’s New Risk Factor After FSKR Merger
WM Technology Snaps Up Sprout; Shares Jump









