The U.S. Food and Drug Administration (FDA) has extended the review period for Pfizer (PFE) and Opko Health Inc.’s (OPK) Biologics License Application (BLA) for somatrogon for the treatment of growth hormone deficiency (GHD) in pediatric patients.
Since Pfizer submitted additional data to the original BLA, the FDA extended the Prescription Drug User Fee Act (PDUFA) goal date by three months to January 2022. Florida-based Opko Health is a multinational biopharmaceutical and diagnostics company. (See Opko stock chart on TipRanks)
The companies signed an agreement for the global development and commercialization of somatrogon for the treatment of GHD in 2014. As per the terms of the agreement, Opko conducts the clinical program and Pfizer is responsible for registering and commercializing the product for GHD.
Somatrogon has Orphan Drug designation in the European Union and the U.S. for the treatment of GHD in adults and children. Opko’s shares closed 3.4% down at $3.75 on Friday, while Pfizer’s shares slipped only 0.6% to close at $43.94. (See Pfizer stock chart on TipRanks)
Following the announcement, Piper Sandler analyst Edward Tenthoff reiterated a Buy rating on Opko with a price target of $5 (33.3% upside potential). Despite the delay, the analyst is confident about the approval of somatrogon on the back of positive Phase III data.
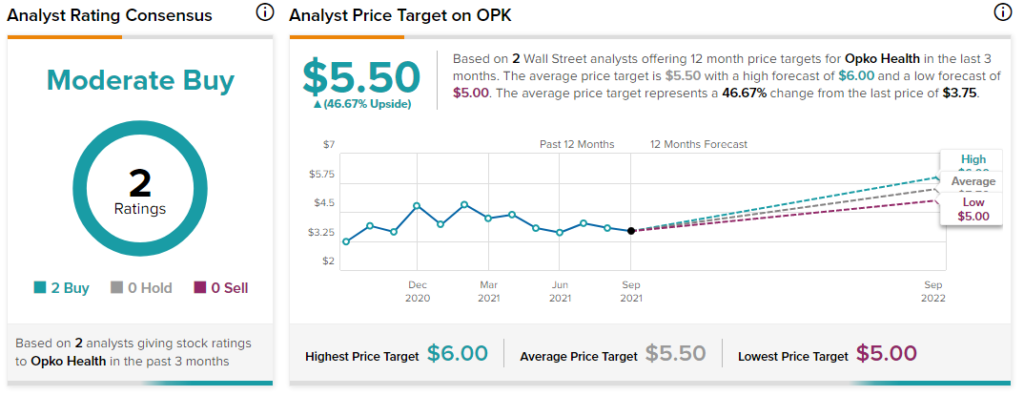
Overall, the stock has a Moderate Buy consensus rating based on 2 Buys. The average Opko Health price target of $5.50 implies 46.7% upside potential. Shares have gained nearly 18% over the past year.
Recently, Citigroup analyst Andrew Baum maintained a Hold rating on Pfizer with a price target of $43 (2.1% downside potential). In a research note to investors, the analyst said, “Investors should remain upbeat over the outlook for Pfizer’s COVID-19 vaccine revenues.”
Overall, the stock has a Hold consensus rating based on 2 Buys and 9 Holds. The average Pfizer price target of $45.55 implies 3.7% upside potential. Shares have gained 27.5% over the past year.

Related News:
Middleby Acquires Imperial Commercial Cooking Equipment
Roku Introduces Streaming Player Line-Up in Germany
Vertex Snaps Up LCR-Dixon, Enhances Global SAP Tax Offerings









