Trailing Pfizer (PFE) by just one day, Moderna, Inc. (NASDAQ: MRNA) has successfully applied for U.S. FDA emergency use authorization for its updated COVID-19 booster shot, mRNA-1273.222. The biotechnology company is seeking approval for a 50 µg booster dose for adults 18 years of age and older.
The updated vaccine targets the original strain of SARS-CoV-2, along with both subvariants of the Omicron strain, BA.4/BA.5, which are believed to be presently dominant in the United States.
The vaccine developer is aiming to receive authorization before the U.S. government’s fall booster campaign.
According to a report published by The Wall Street Journal, the U.S. government aims to provide 175 million updated vaccine doses to states, pharmacies, and other vaccination sites as a part of its fall COVID-19 booster campaign, which is to be launched in September. A Reuters report mentioned that Britain and some European Union members are also working on similar campaigns as a precaution measure.
Moderna’s application is based on preclinical data and the data from a clinical trial, which it conducted for the booster candidate targeting BA.1 Omicron variant. Notably, a Phase 2/3 trial for the mRNA-1273.222 has already begun. If approved, Moderna will have the mRNA-1273.222 shipments ready for delivery in September.
What Is MRNA Stock’s Price Forecast?
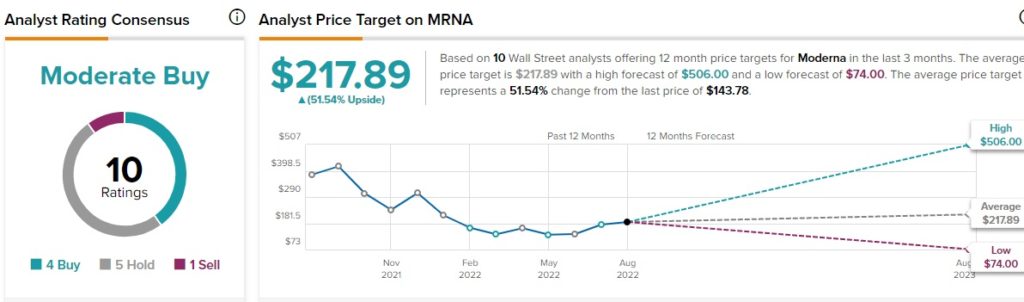
Moderna’s average price forecast is $217.89, which implies 51.5% upside potential to current levels. On TipRanks, analysts have a Moderate Buy consensus rating on Moderna, which is based on four Buys, five Holds, and one Sell.
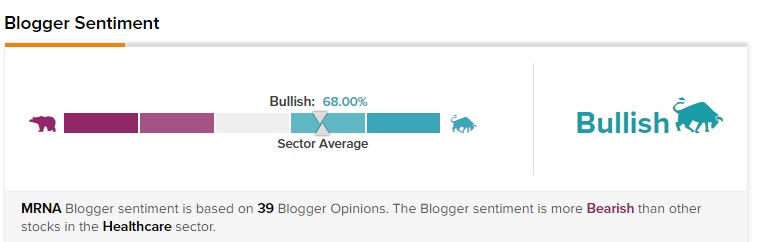
Further, financial bloggers are 68% bullish on Moderna stock, largely in line with the sector average of 69%.
Final Thoughts
Moderna has been delivering impressive financial performance based on solid COVID-19 vaccine sales. The biotechnology company recently announced that it signed advance purchase agreements worth about $21 billion with several countries for its COVID-19 vaccine and booster doses. This should help Moderna further boost its top-line numbers.
Read full Disclosure.