The U.S. Food and Drug Administration has expanded the emergency use authorization for AbCellera Biologics’ (ABCL) bamlanivimab, which is administered with etesevimab to prevent SARS-CoV-2 infection or symptomatic COVID-19.
Following the news, shares of the biotechnology firm rallied 19.8% in Thursday’s extended trade. The stock further gained 16.7% in pre-market trading session on Friday.
The expanded authorization is based on the Phase 3 BLAZE-2 trial that showed bamlanivimab helped in preventing COVID-19 and reducing the risk of contracting the disease by up to 80% in nursing home residents and up to 57% among residents and staff of long-term care facilities.
Notably, bamlanivimab and etesevimab together are seen to retain neutralization activity against the Alpha and Delta variants. (See AbCellera stock chart on TipRanks)
The CEO and President of AbCellera, Carl Hansen (Ph.D.), said, “With this expanded authorization, these antibodies, which have been shown to be effective against the highly contagious Delta variant, can now be used to protect some of the most at-risk people exposed to the virus.”
On September 15, Berenberg Bank analyst Gal Munda reiterated a Buy rating on AbCellera with a price target of $48. The analyst’s price target implies upside potential of 142.4% from current levels.
Munda anticipates AbCellera to report a loss of $0.01 per share for the third quarter of 2021.
Consensus among analysts is a Strong Buy based on 3 unanimous Buys. The average AbCellera price target of $36.50 implies upside potential of 84.3% to current levels.
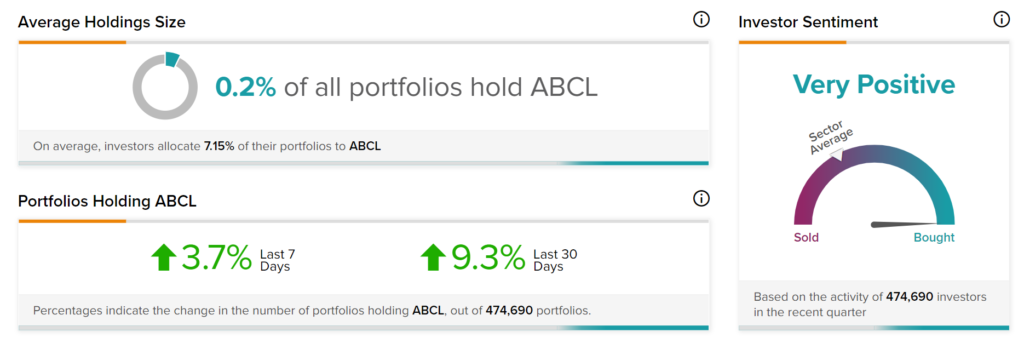
TipRanks’ Stock Investors tool shows that investors currently have a Very Positive stance on AbCellera with 9.3% of investors on TipRanks increasing their exposure to ABCL stock over the past 30 days.
Related News:
Diamondback to Accelerate Capital Return; Announces Share Buyback Program
Profire Energy Approves Share Repurchase Program
Johnson Controls Issues $500M Sustainability-Linked Bond









