Australia has announced a $1.2 billion supply and production agreement for over 84.8 million Covid-19 vaccine doses with the University of Oxford/AstraZeneca (AZN) and the University of Queensland/CSL
According to the government, this will enable it to offer a free COVID-19 vaccine progressively throughout 2021 in Australia, if ongoing trials prove successful. It wants Australians to have access to 3.8 million doses of the University of Oxford vaccine in January and February.
Prime Minister Scott Morrison said both vaccines would need to be proven safe and effective, and meet all necessary regulatory requirements, prior to being made available to the public.
“By securing the production and supply agreements, Australians will be among the first in the world to receive a safe and effective vaccine, should it pass late stage testing,” the Prime Minister said.
“There are no guarantees that these vaccines will prove successful, however the agreement puts Australia at the top of the queue, if our medical experts give the vaccines the green light.”
Both agreements allow for additional orders to be negotiated and for doses to be donated or on-sold (with no mark-up) to other countries or international organizations, the government added.
The University of Queensland (UQ) recently announced that pre-clinical testing showed the vaccine is promising and already effective in animal models.
Meanwhile, the University of Oxford/AstraZeneca vaccine candidate AZD1222 has now entered Phase three trials. To date it has generated strong immune responses, with no significant safety concerns. Results from the late-stage trials are anticipated later this year, depending on the rate of infection within the clinical trial communities.
The vaccine uses a replication-deficient chimpanzee viral vector based on a weakened version of a common cold virus (adenovirus) that causes infections in chimpanzees and contains the genetic material of the SARS-CoV-2 virus spike protein. After vaccination, the surface spike protein is produced, priming the immune system to attack the SARS-CoV-2 virus if it later infects the body.
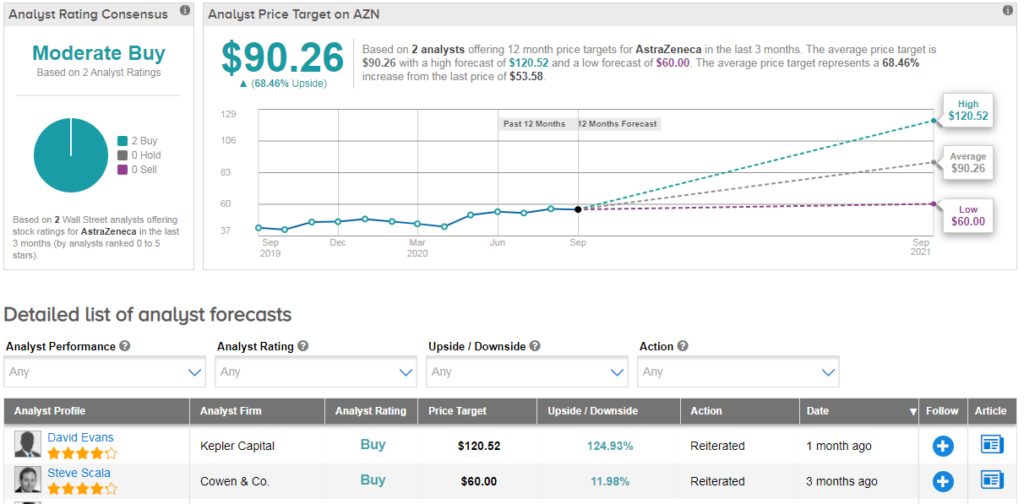
AZN shares have gained 7% this year as the drugmaker joined the list of companies engaged in the development of a potential coronavirus vaccine. Looking ahead, the $90 average analyst price target puts the upside potential at a promising 68% in the coming 12 months.
Overall, the stock scores a Moderate Buy consensus from the analyst community based on 2 recent Buy ratings from Kepler Capital and Cowen & Co. (See AstraZeneca stock analysis on TipRanks).
Related News:
Morgan Stanley Turns Bullish On Eli Lilly, Lifts PT
Moderna Slips 4% Amid Report Of Slowdown In Covid-19 Trial Enrollment
Is Trevena Stock a Buy Right Now? This Is What You Need to Know









