According to a report published by Reuters, the U.S. Food and Drug Administration is seeking a federal investigation into the interactions between its representatives and Biogen (BIIB) that led to the approval of the company’s Alzheimer’s disease treatment Aduhelm last month.
Massachusetts-based Biogen is a multinational biotechnology company that develops, manufactures and sells therapies for the treatment of neurodegenerative and neurological diseases.
Biogen’s shares closed 3% lower on Friday after Janet Woodcock, the FDA’s Acting Commissioner, asked the Office of the Inspector General at the Department of Health and Human Services (HHS) to check whether the talks were consistent with the agency’s policies.
The spokesperson of HHS’s Office of the Inspector General, Tesia Williams, said, “We received the letter and are reviewing it for appropriate action.” (See Biogen stock chart on TipRanks)
The FDA approved Aduhelm on June 7 despite strong objections from its experts. Following the approval, Woodcock said, in a letter posted on Twitter (TWTR), “There continue to be concerns raised… regarding contacts between representatives from Biogen and FDA during the review process.”
Stifel analyst Paul Matteis recently reiterated a Buy rating on Biogen with a price target of $443 (23.7% upside potential). The analyst said the actual content of Woodcock’s letter “doesn’t seem to pique questions in a way that would actually impact Aduhelm’s marketing authorization.”
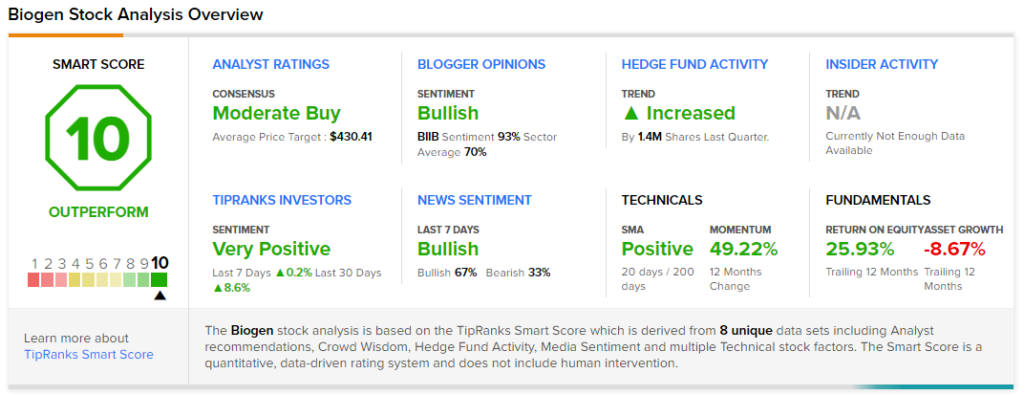
Overall, the stock has a Moderate Buy consensus based on 15 Buys and 11 Holds. The average Biogen price target of $430.41 implies 20.2% upside potential. The company’s shares have gained 47.4% year-to-date.
According to TipRanks’ Smart Score rating system, Biogen scores a “Perfect 10,” suggesting that the stock is likely to outperform market averages.
Related News:
Altria to Sell Ste. Michelle Wine Estates Business for $1.2B; Shares Rise
MTY Food Swings to a Profit in Q2, Restores Dividend; Shares Gain Almost 15%
Brunswick Corporation’s Freedom Acquires Spain’s Fanautic Club









