Clinical-stage biopharmaceutical company atai Life Sciences N.V. (Nasdaq: ATAI) revealed that it has bagged clearance from the U.S. Food and Drug Administration (FDA) to conduct a clinical DDI study of PCN-101, designed to treat mental health disorders.
The company seeks to begin the study early in 2022 through its platform company Perception Neuroscience.
atai Life Sciences expects that PCN-101 carries some unique properties that are different from currently available antidepressants and has the potential to address key patient needs, including the potential of rapid action and anti-suicidal effect.
The company said that the clinical trial would be conducted alongside an existing Phase 2a proof-of-concept trial in treatment-resistant depression, recently initiated in Europe. Moreover, to make the treatment self-administered, atai Life Sciences plans to run a bioavailability study in 2022 to bridge the IV formulation to a subcutaneous formulation of PCN-101.
The CEO and Co-Founder of atai Life Sciences, Florian Brand, said, “We see great promise in PCN-101 as a potentially rapid-acting anti-depressant with a more favorable safety and tolerability profile than S-ketamine, which could enable at-home use.”
Price Target
Recently, RBC Capital analyst Brian Abrahams maintained a Hold rating on the stock with a price target of $12 (80.2% upside potential).
Overall, the rest of the Street has a bullish outlook on the stock with a Strong Buy consensus rating based on 7 Buys and 1 Hold. The average atai Life Sciences price target of $26.57 implies upside potential of about 299%.
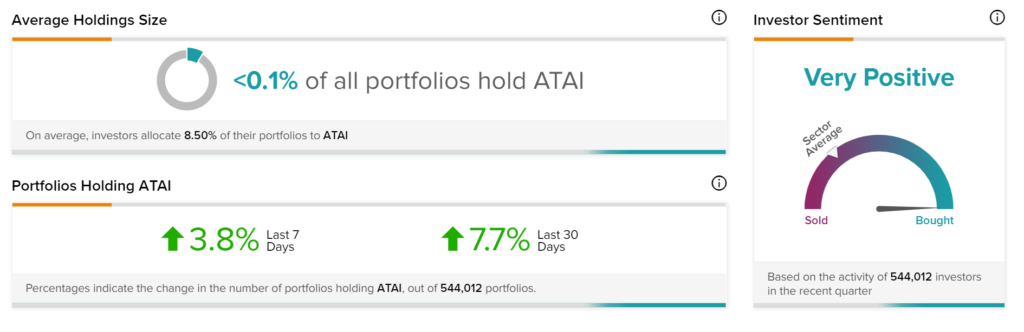
Positive Sentiment
TipRanks’ Stock Investors tool shows that investors currently have a Very Positive stance on ATAI with 7.7% of investors on TipRanks increasing their exposure to the stock over the past 30 days.
Download the TipRanks mobile app now
To find good ideas for stocks trading at attractive valuations, visit TipRanks’ Best Stocks to Buy, a newly launched tool that unites all of TipRanks’ equity insights.
Read full Disclaimer & Disclosure
Related News:
Coinbase Acquires Crypto-Derivatives Trading Platform FairX
Ares Capital to Offer 10M Common Stock; Shares Fall
Bionano Reveals Preliminary 2021 Results; Shares Rise









