Let’s talk about the economy and COVID-19. Normally, this is a subject that will dwell on the economic disaster of the past year – the sharp recession last winter, the huge spike in unemployment, the unprecedented surge in government debt. All of these are important, even urgent, topics for discussion, but we’re here to talk about vaccines and opportunity.
Specifically, the vaccines coming out of the biotech sector, and the opportunities they’ll bring. The rapid spread of COVID around the world led to huge push by the Big Pharma industry to create a working vaccine for the disease – a push that has had successful results. Two working vaccines were in production and getting to the general public before the end of 2020, and several others vaccine manufacturers had products in development or in approval in the early months of this year. It was an impressive display of medical and technological know-how, put to the public benefit.
The advent of vaccines, however, did not put the COVID pandemic to rest. As this virus has been mutating, and new variants are beginning to spread. The existing vaccines may prove effective, but they may not – and just as we get a new flu shot every year, we may be looking at a situation in which we will need our regular ‘corona boosters’ to cope with the shifting variants of the virus.
And now we get to the opportunities. A global vaccination program, and new vaccines for new strains, will present a huge business opening for biopharma companies involved in vaccine research and production. Using the TipRanks database, we’ve pulled the details on three biopharma firms that stand to gain as the vaccination opportunities expand.
Novavax, Inc. (NVAX)
We’ll start in the DC suburbs, where Gaithersburg, Maryland-based Novavax is a specialist biotech firm focusing on developing vaccines for respiratory illnesses. The company was involved in earlier work on the SARS and MERS viral outbreaks, which left it uniquely pre-adapted for work on the COVID-19 agent; both the SARS and MERS viruses come from the same class of coronaviruses as COVID-19.
Earlier this month, Novavax reported that its COVID-19 vaccine program had completed Phase 3 trials, and demonstrated a 96.4% efficacy against the original strain of the virus. In addition, Novavax’s vaccine is also in a Phase 2b trial against the South Africa variant of the virus. The company announced that data from that trial shows a 55.4% efficacy against the South African strain, in patients who are HIV-negative. In both studies, the vaccine candidate achieved its statistical goals, including 100% protection against severe disease and hospitalization.
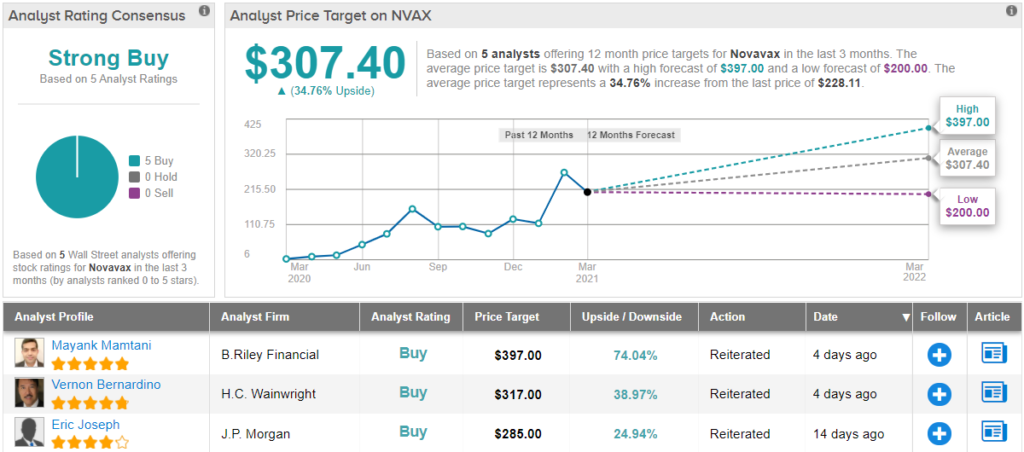
Covering this stock for B. Riley Securities, 5-star analyst Mayank Mamtani notes the company’s successful vaccine trials, and sees them as the surest path forward for Novavax.
“NVAX [is] ahead in the marathon of building durable vaccines’ franchise, primarily via designing multi-valent booster vaccine candidate that could also include both their late-stage vaccine candidates against C-19 and influenza,” Mamtani noted.
The analyst added, “We continue to believe recent stock volatility presents yet another attractive opportunity to accumulate NVAX shares, particularly in light of a highly derisked catalyst profile constituting of several regulatory approvals globally beginning with reiterated guidance of U.K. MHRA filing in early 2Q and U.S. FDA filing within 2Q.”
To this end, Mamtani rates NVAX shares as a Buy, and his $397 price target implies a 74% upside potential for the coming year. (To watch Mamtani’s track record, click here)
Overall, Wall Street analysts agree that NVAX is a solid prospect for investors; all 5 of the recent reviews here are to Buy, making the Strong Buy consensus rating unanimous. Shares are trading for $201.50, and the $307.40 average price target suggests ~35% one-year upside. (See NVAX stock analysis on TipRanks)
VBI Vaccines (VBIV)
The next COVID warrior we’re looking at is VBI Vaccines, another company that specializes in vaccine research and has taken up the challenge of the novel coronavirus. The company has a history of working with difficult to treat, chronic viral diseases, such as Hepatitis B, but has also worked with other coronaviruses in the past.
The big news here came out earlier this month, when VBI and CEPI (Coalition for Epidemic Preparedness Innovations) together announced a partnership to use VBI’s vaccine candidates in research against the B.1.351 variant of the COVID-19 virus. This is the South African variant, and is known to be more resistant to existing treatments. Development of a vaccine against this strain would be a major step forward against the COVID-19 pandemic.
The partnership comes with $33 million in funding for VBI adapt its enveloped virus like particle (eVLP) vaccine technology against the South African variant.
VBI’s eVLP technology has already been tested in preclinical studies against SARS-CoV-2 (the COVID-19 agent), and has shown promise in inducing antibody titers against all three variants. Additionally – and, perhaps, more excitingly – the vaccine candidate has also shown reactivity to HCoV-OC43, one of the already known seasonal coronaviruses.
All of this has attracted attention from Raymond James analyst Steven Seedhouse.
“Partnership with CEPI coupled with our macro thesis on COVID (which is that the virus is endemic and variant vaccines plus annual boosts will be required for years or even in perpetuity) collectively sets up VBI for a meaningful elevation in profile of its COVID vaccine programs and more broadly its eVLP platform with an important organization (CEPI) that also announced a $3.5B initiative today to cut vaccine development timelines for future pandemics,” Seedhouse opined.
Seedhouse puts a $9 price target on this stock, showing confidence in a robust 159% upside for the next 12 months, and supporting his Strong Buy rating. (To watch Seedhouse’s track record, click here)
Overall, we’re looking at a stock with a unanimous Wall Street analyst consensus – 4 reviewers have weighed in, and they have all put a seal of approval here, for a Strong Buy rating. VBIV shares are trading for $3.47, and the $6.75 average price target suggests room for 94% growth this year. (See VBIV stock analysis on TipRanks)
Moderna, Inc. (MRNA)
Moderna has made waves in the news, as it was one of the first two companies, along with Pfizer, to release an effective COVID-19 vaccine to the market. Moderna’s shot – a two-dose vaccine based on mRNA technology – has been in the headlines since November, and the company’s stock skyrocketed 480% over the past 12 months
From the beginning of the vaccination programs, there have been questions and concerns relating to the possibility of booster shots. Will they be needed, will they be safe, will they increase efficacy? Moderna is working to answer those questions, and on March 10 the company announced that it has begun dosing patients for its study of COVID boosters, in an amendment to ongoing Phase 2 trials. The stock is up more than 9% since that announcement.
Moderna’s booster announcement highlights an unusual feature of the COVID vaccine development program, one that is visible across the board. The vaccines are moving fast through the development process. Partly, this is due to the ability of companies to build on previous work in relation to SARS and MERS, and partly it is due to development programs like Moderna’s pushing the vaccine candidates through separate parts of the development and clinical trial phases simultaneously. Moderna’s booster program will test the current vaccine, and vaccine candidates, against several strains of the novel coronavirus.
In a note for Brookline Capital, analyst Leah Rush Cann sees Moderna on a long-term runway to increased sales and profits, deriving directly from the company’s success with the COVID-19 vaccine program.
The analyst writes that the “announcement for progress on a variant-addressing COVID-19 vaccine supports our conviction for our outlook that mRNA-1273, and potentially variant-addressing vaccines or boosters, could have 2030 sales of $51.4 billion and account for 51% of Moderna’s 2030 revenue…”
The analyst added, “We continue to believe that the Moderna COVID-19 vaccine has increasingly superior attributes, and re-confirms the flexibility and power of mRNA platforms; allowing them to adapt for variations and mutations in infectious diseases very quickly.”
In line with her comments, Cann set her outlook on the stock to Buy, and her $205 target price suggests that the growth will hit 32% over the next 12 months. (To watch Cann’s track record, click here)
Debates over the vaccines have been reflected in debates over the stock, and Moderna has a mix of reviews from Wall Street’s analysts. Of the 13 reviews on file, 6 are to Buy, 5 to Hold, and 2 to Sell, making the consensus rating a Moderate Buy. The average price target is $168.08, implying an upside of 8% from the trading price of $155. (See MRNA stock analysis on TipRanks)
To find good ideas for stocks trading at attractive valuations, visit TipRanks’ Best Stocks to Buy, a newly launched tool that unites all of TipRanks’ equity insights.
Disclaimer: The opinions expressed in this article are solely those of the featured analysts. The content is intended to be used for informational purposes only. It is very important to do your own analysis before making any investment.