Shares of Adial Pharmaceuticals, Inc. (NASDAQ: ADIL) soared by more than 50% in pre-market trading on Tuesday after the clinical-stage biopharmaceutical company gave a positive regulatory update for AD04, the company’s lead compound for the treatment of Alcohol Use Disorder (AUD).
The company stated that the U.S. FDA had confirmed a Type C meeting in the second quarter of this year to discuss the clinical program. ADIL also had meetings scheduled or planned with five European country-level regulatory authorities including France, Sweden, Finland, the U.K., and Germany. The company was also in advanced discussions with partners in the U.S. and Europe.
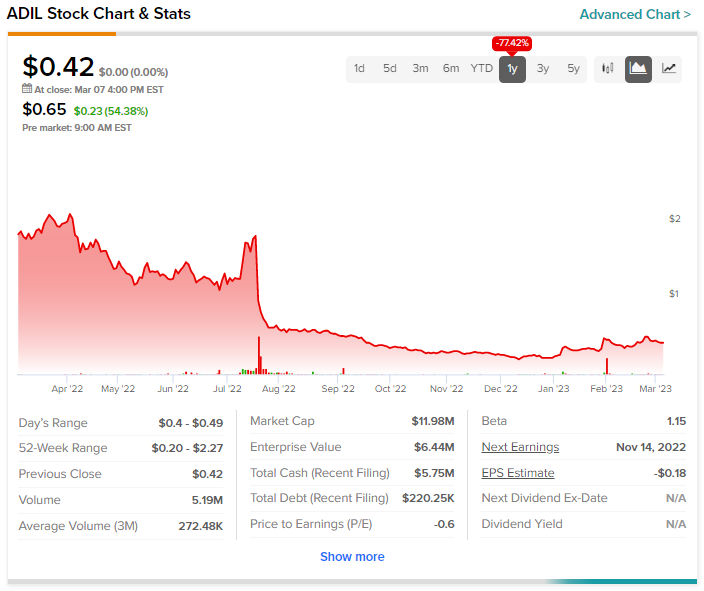
ADIL stock has plunged by more than 75% in the past year.









